People with chronic inflammatory diseases are taking part in a new cell therapy clinical trial that one participant said made them feel “miles better”.
The POLARISE trial, being organised by BHP founder-member the University of Birmingham and funded by a grant from Innovate UK is testing a type of cell therapy – stromal cells – to see whether they can resolve symptoms and inflammation in patients with certain autoimmune diseases including rheumatoid arthritis and primary sclerosing cholangitis.
A Phase 2 trial, POLARISE will investigate the safety and activity of ORBCEL – a stromal cell therapy that has been developed by Orbsen Therapeutics Ltd. Stromal cells are rare cells found naturally in the human body where they stimulate resolution of injury and inflammation via a natural healing process called efferocytosis. Stromal cells are also allogeneic – which means they can be purified from one donor and given to multiple patients without causing allergic reactions – so there is no need for donor matching.
These rare stromal cells are ethically sourced and purified from human donor tissue and expanded to therapeutic doses at the University of Birmingham’s Medicines Manufacturing Facility (MMF).
The ORBCEL therapy is administered intravenously across two visits with subsequent hospital appointments to check on the progress of their condition during a two-year trial period.
Philip Newsome, Professor of Hepatology and Honorary Consultant Hepatologist at the University of Birmingham is leading the POLARISE trial, and explained: “Stromal cells are an exciting potential treatment for inflammatory diseases. These diseases are debilitating and very hard to treat as the body has switched a natural defence system for dealing with threats to one that starts attacking itself. It’s therefore critical to find ways to support the body to naturally deal with inflammation rather than turn off the defences which can lead to all sorts of infections. Early results from previous trials using Orbsen’s ORBCEL stromal cell therapy are encouraging and we’re hopeful that the treatment will be beneficial for some patients.”
Stromal cells such as Orbsen’s ORBCEL therapy can be purified from bone marrow or umbilical cord tissues donated by healthy individuals with donor consent under ethical approval by the Anthony Nolan Trust. While each single bone marrow or umbilical cord contains only few thousand stromal cells – these cells can be purified by Orbsen’s technology to undergo controlled expansion in cleanroom bio-reactors to produce a thousand allogenic doses of ORBCEL from each tissue.
Within the Innovate UK-funded Advanced Therapies Treatment Centre (ATTC) Consortium and POLARISE trial – these tissues are transported from the Anthony Nolan centres to the Advanced Therapies Facility (ATF) at the University of Birmingham – where Orbsen and ATF staff collaborated to purify and manufacture doses of Orbsen’s Stromal cell therapy – ORBCEL- using patented technologies and Terumo’s Quantum Cell Expansion Bioreactors.
Orbsen Therapeutics Chief Scientific Officer, Steve Elliman said: “We are delighted to continue our significant and productive clinical collaborations with Prof. Newsome, the University of Birmingham – and the Anthony Nolan Trust – to determine the safety and efficacy of our ORBCEL therapies in patients with chronic inflammatory diseases.
“These First in Human (FIH) trials are difficult to undertake and deliver – even more so during the COVID19 pandemic. These trials are not possible without brave patients – like Hannah Dines – who volunteer to participate in these rigorous safety trials. And so, we take this opportunity to thank the patients, nurses and clinical teams who work so hard to complete these invaluable studies.
“We look forward to completing these important safety trials and look forward to examine how ORBCEL can encourage resolution of symptoms in patients with chronic inflammatory disease.”
The Innovate UK-funded POLARISE trial represent the third major clinical trial collaboration between The University of Birmingham and Orbsen Therapeutics to assess the safety and efficacy of Orbsen’s ORBCEL therapy. Professor Phil Newsome is also leading the EU FP7 funded MERLIN clinical trial that is assessing ORBCEL as a therapy for patients with autoimmune liver diseases. The MERLIN trial is complete and is expected to report in the first half of 2024.
Orbsen is also collaborating with Prof Paul Cockwell at the University of Birmingham and Professor Giuseppe Remuzzi at the Mario Negri to assess the safety of ORBCEL as a therapy for Chronic Kidney Disease caused by Type 2 diabetes, in a Phase 1/2 clinical trial called NEPHSTROM. Professors Cockwell and Remuzzi recently published the first results from NEPHSTROM in the prestigious Journal of the American Society of Nephrology (JASN). In the NEPHSTROM trial publication in JASN, a low dose of ORBCEL was reported to be safe and promote stabilization of kidney function over 18 months in patients suffering with Progressive Chronic Kidney Disease and type 2 diabetes.
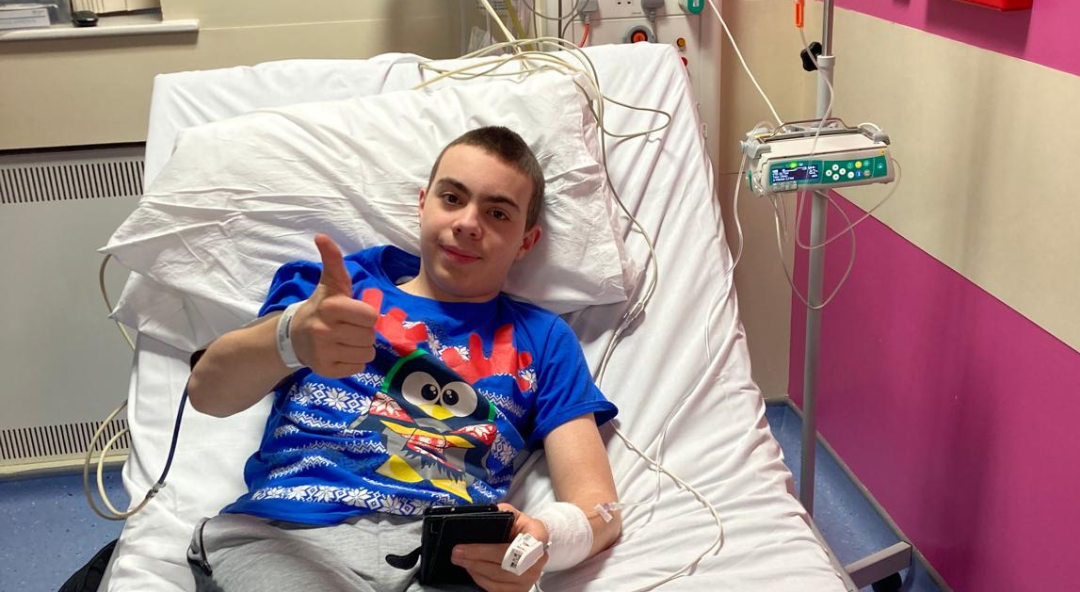
Patient story – Hannah Dines, Rio 2016 Paralympian
Self-confessed ‘type A person’, Hannah Dines is one for setting mad goals. Born with cerebral palsy, freelance writer and sportswoman Hannah trained and raced for Great Britain in para-cycling including racing at the Rio 2016 Paralympic games, and now represents GB in adaptive surfing.
However, in 2021 during the buildup to the delayed Tokyo games Hannah was diagnosed with a chronic inflammatory disease called Primary Sclerosing Cholangitis (PSC) in which the bile ducts in the liver get progressively narrower can lead to liver failure and impacts other organs like the spleen, intestines and bowel.
Hannah explains: “I was diagnosed with PSC after struggling with major fatigue and worsening of my spasticity from my cerebral palsy. I would train and feel very ill but do it anyway. I love moving my body and during the training I still felt that joy. Still, I began to fear the symptoms that would come after. I used to call it having an ‘exercise hangover’ though I rarely drank alcohol and was in my twenties. I would ensure I had at least four hours after training to collapse in bed, too tired to even watch TV, feeling too ill to sleep, known as malaise.”
“By the point of diagnosis though I was really ill and sleepy every day, I couldn’t focus but I kept pushing with my training, a part time job and then bed. Finally, a clinical doctor took my blood to put me on an alternative spasticity medication that required a liver function test. That’s when I was sent to a liver clinic and they took a liver biopsy right away and found out my sclerosis was pretty serious.
“It made all my symptoms make sense and because I was young and sporty no-one misdiagnosed me with fatty liver or alcoholism, which was nice, even if it didn’t really lessen the impact of having PSC.”
After receiving her diagnosis, qualified physiologist Hannah knew she wanted to try and take part in a clinical trial although received a series of rejections due to the advanced nature of her PSC.
Hannah continues: “I was recommended for POLARISE and I didn’t hesitate. The day after my first dose I felt incredible and not just because the clinicians administering the drugs were so nice. This effect lasted a couple of days and I truly felt released from PSC.”
“I was still competing at a sport: adaptive surfing and I booked all my contests because I knew I wouldn’t need to cancel. I laughed out loud on an aeroplane because I felt real energy for the first time in years. It was probably the steroids or a placebo effect but my liver function tests also got much better.
“My second dose was a little underwhelming compared to my first, but I still felt miles better. My “malaise” and feeling kind of “dead” had gone away.
“I used to obsess over my blood values and stopped checking them. I started setting goals more than two months in advance, which I had decided not to do after a year of having to cancel everything. We’re now six months and I still big hits of malaise but just to know that respite might be possible like at the start of my trial…that’s really special..
“All I can do is hope the findings are positive and this can become a regular treatment for people with PSC. No matter the result of POLARISE it has given me real hope for the future.”
Not letting PSC stop her, Hannah has taken up adaptive surfing and last year represented GB at the world championships, finishing fourth in her category and supporting Team GB to their most successful championships yet.
You might also be interested in: